The study found strong statistical differences between patients who received Nexalin’s DIFS® technology in combination with escitalopram (Lexapro) and a placebo group who received escitalopram alone.
Two-thirds of patients who received the combination treatment saw improvement.
compared with only one-third of patients receiving medical therapy alone.
HOUSTON, June 26, 2024 (GLOBE NEWSWIRE) — Nexalin Technology, Inc. (Nasdaq: NXL; NXLIW) (the “Company” or “Nexalin”) today announced positive results from a clinical trial designed to evaluate the feasibility, safety and efficacy of transcranial alternating current dynamic frequency stimulation (tACS) as an add-on treatment for symptoms of Major Depressive Disorder (MDD), also known as clinical depression, a psychiatric illness that affects mood, behavior, appetite and sleep.
The study was designed as a 4-week, double-blind, randomized, sham-controlled trial, recruiting 66 participants who were randomly assigned to receive 20 40-minute sessions of either real treatment or sham stimulation using the company’s Non-Invasive Deep Intracranial Frequency Stimulation (DIFS®) technology (77.5Hz, 15mA). Throughout the 4-week period, participants were treated with escitalopram (Lexapro), a selective serotonin reuptake inhibitor (SSRI). Escitalopram is a medication commonly prescribed to treat anxiety disorders and severe depression.
A significant difference was observed in the reduction of Hamilton Depression Rating Scale (HAMD-17) scores at week 4 (t = 3.44, P = 0.001). The response rate at week 4 was significantly higher in the active tACS group than in the sham tACS group (22 of 33 patients). [66.7 %] 11 of 33 [33.3 %]P = 0.007). In the active tACS group, a correlation was found between the mean change in alpha power and HAMD-17 scores at week 4 (r = 2.38, P = 0.024), and the mean change in alpha power was significantly greater in responders (Z = 2.46, P = 0.014). No serious adverse events were observed in this study.
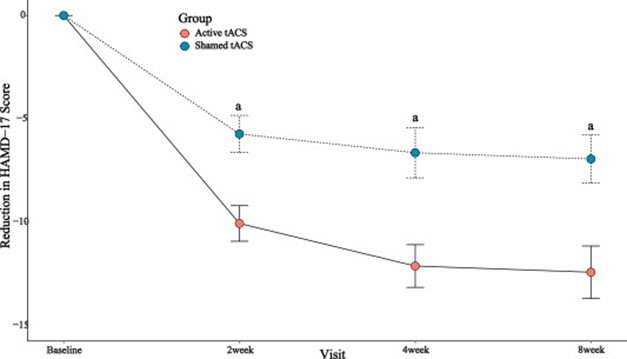
The findings were published in Brain Stimulation, the premier peer-reviewed journal in the field of neuromodulation.Add-on effect of transcranial alternating current stimulation (tACS) in major depressive disorder: a randomized controlled trialThey conclude that “the antidepressant effects of tACS are significant, and the combination of tACS with antidepressants is a feasible and effective approach for the treatment of MDD.”
Dr. David Owens, Chief Medical Officer at Nexalin, commented, “We are proud to report these positive results and honored to have them published in such a prestigious peer-reviewed journal. While we have previously demonstrated strong treatment efficacy with our DIFS technology alone, this is the first study to evaluate our technology in combination with an SSRI, the gold standard treatment in the United States. Most notably, the study showed strong, statistically significant differences between the active group receiving DIFS plus medication and the placebo group receiving medication only – exactly what we hoped to show in this trial. In fact, only one-third of treated patients receiving medication alone saw improvement, compared to two-thirds of patients receiving the combination. Also notable was the lack of significant side effects reported.”
“We believe this data strongly supports the growing body of clinical evidence supporting the potential of Nexalin’s new advanced 15mAmp waveform to help combat the ongoing global mental health epidemic, either as a standalone drug-free treatment or in combination with existing drug therapies,” said Mark White, CEO of Nexalin. “According to the 2022 National Survey on Drug Use and Health, approximately 22.47 million people, or 8.8% of U.S. adults, have experienced at least one severe depressive episode. Additionally, according to Future Market Insights, the MDD treatment market is projected to reach $14.96 billion by 2032, up from $11.51 billion in 2022. We believe this data is further evidence that our non-invasive, drug-free device is making a significant impact in improving mental health care outcomes for patients with MDD. We look forward to advancing our technology to provide new and effective treatments to millions of patients suffering from mental health issues in the U.S. and around the world.”
About Nexalin Technology, Inc.
Nexalin designs and develops innovative neurostimulation products that uniquely help to combat the ongoing global mental health epidemic. All of Nexalin’s products are believed to be non-invasive and undetectable to the human body and are developed to provide relief to people suffering from mental health issues. Nexalin utilizes bioelectronic medical technology to treat mental health issues. Nexalin believes its neurostimulation medical devices can penetrate deep structures in the midbrain that are associated with mental health disorders. Nexalin believes that the deeper penetrating waveforms of its next generation device will enhance patient response without side effects. The Nexalin Gen-2 15 milliamp neurostimulation device has been approved in China, Brazil and Oman. Additional information about the company can be found at https://nexalin.com/.
Forward-Looking Statements
This press release contains statements that constitute “forward-looking statements.” These statements relate to future events or Nexalin’s future financial performance. Any statements that refer to expectations, projections or other characterizations of future events or circumstances or that are not statements of historical fact (including, but not limited to, statements that Nexalin or its management team “believes,” “expects,” “anticipates,” “plans,” “intends” or similar expressions) should be considered forward-looking statements that involve risks and uncertainties that may cause actual events or Nexalin’s actual results to differ materially from those indicated in the forward-looking statements. Forward-looking statements are subject to many conditions, many of which are beyond the Company’s control, including those described in the Risk Factors section of our Report on Form 10-K for the year ended December 31, 2023 and other filings with the Securities and Exchange Commission. Copies of such filings may be obtained from the SEC’s website. Click here for detailsThese forward-looking statements speak only as of the date hereof and may become outdated over time, and the Company undertakes no obligation to update these statements with revisions or changes after the date hereof, except as required by law.
contact:
Crescendo Communications LLC
Phone: (212) 671-1020
Email: inquiry
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/ca5f1a24-dc0a-4bb1-9bd1-86203176a408


